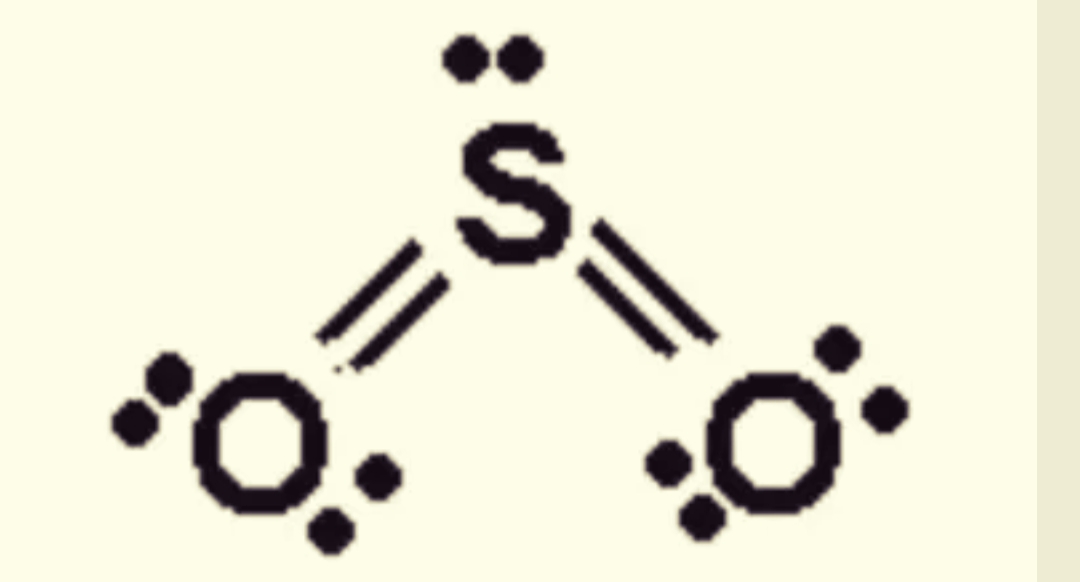
Typical weaknesses include poor cycle stability, unimpressive rate performance and unsatisfactory energy production due to the dissolved polysulfide intermediates (Li 2S x, 4 ≤ x ≤ 8) in the electrolyte, and the low electrical and ionic conductivity of elemental sulfur and its final discharge product. 1–5 Nevertheless, performances of Li–S batteries are not satisfactory as in theory. Lithium–sulfur (Li–S) batteries have become a hot topic due to their excellent specific capacity (1675 mA h g −1) and theoretical energy density (2600 W h kg −1). Introduction With the rapid development of electronic vehicles and large-scale power transfer installations, the demand for sustainable batteries with high energy density is increasing. Several important electrochemical reactions also have been verified by cyclic voltammetry curves. The inclined line arises from the diffusion process in the detectable potentials and frequency range. Based on detailed analysis of the change in kinetic parameters obtained from simulating the experimental EIS data as functions of potential, the high-frequency, middle-frequency and low-frequency semicircles/arcs can be attributed to the Schottky contact reflecting the electronic properties of materials, the charge transfer step and the formation of Li 2S respectively. An appropriate equivalent circuit is proposed to fit the experimental EIS data. In the second discharge region (2.00–1.70 V), where soluble polysulfides are reduced to Li 2S, the EIS spectra exhibit three semicircles/arcs as the frequency decreased. Under some circumstances, protons and electrons can be converted to other particles in certain nuclear reactions, but in doing so, the net charge for the reactions is zero.Electrochemical impedance spectroscopy (EIS) was used to study the initial discharge/charge process in a sulfur cathode with different potentials. Note: While it is a good model to think of conservation as an inability to increase or decrease the total number of protons and electrons, it technically isn't 100% accurate. These are referred to as the elementary charge. 
To give a brief quantitative overview of electric charge, the unit for charge is the Coulomb, denoted by "C". If a baseball is thrown upwards at an initial kinetic energy, #E_k#, the gravitational potential energy, #E_"PE"#, will be equal to #E_k#. We usually use this principle in physics when we equate the initial energy of an event to the final energy of an event. Another common conservation principle is energy. Here, it is the conservation of mass that is concerned. When you balance chemical equations, you are ensuring that the total number of atoms remain constant throughout the reaction. One way to think about conserved properties is that the total number of protons and electrons in the universe is constant (see Note below).Ĭonservation is a common theme in chemistry and physics. Since protons and electrons are the carriers of positive and negative charges, and they cannot be created or destroyed, electric charges cannot be created or destroyed. Simply put, protons and electrons cannot be created or destroyed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
